Study shows how the brain behaves after gender-affirming surgery in transgender women
Posted September 30, 2019
MRI scans from patients undergoing surgery and hormone treatment show brain adaptations that may be related to gender affirmation
Please read the interview below, in which the researcher Maiko Schneider, member of the INCT Hormona who works at the Department de Psychiatry and Behavioural Neurosciences of McMaster University in Canada, describes the findings of the research that resulted in the article entitled “Effects of Estradiol Therapy on Resting-State Functional Connectivity of Transgender Women After Gender-Affirming Related Gonadectomy” (Schneider MA et al., Front Neurosci. 2019) and that one of the highlights in the recent congress of the American College of Endocrinology.
1) Your study investigated the effect of hormone therapy on the brain of women who underwent gender-affirming surgery. What motivated this investigation?
Originally, I’ve always been very interested in studying the relationship between sexual hormones and mental health, from physiological functioning of hormones to their role on the development of mood and cognitive symptoms. Studying the role of hormones in the transgender population is a unique opportunity to evaluate the role of hormones regardless of the person’s gender, thus enabling to expand our knowledge on neuroendocrinology, which is the field that studies the relationship between hormones and brain. Among post-surgical women, another very important factor should be taken into account: it’s not only about cross-gender hormone therapy but mainly about correcting hypogonadism, which consists on a situation when a person lacks sex hormones, since gender-affirming surgery in our population includes gonadal removal. Therefore, in addition to assessing the effect of cross-gender hormone therapy, this study helped us to better understand the importance of correcting hypogonadism, allowing for the creation of new hypotheses on the functioning of hormones in the brain. Now we gained a little more understanding about the changes that happen in the brain of transgender women when their surgically-induced hypogonadism is corrected.
2) What was the sample for analysis and how did you observe the subjects?
The sample included transgender women who had already undergo gender-affirming surgery at the transdisciplinary gender identity outpatient clinic. We invited transgender women who had undergone the surgery at least one year before and who were willing to interrupt the use of female sex hormones for a certain period and to have their “brain measures” taken using brain MRI scans. After discontinuing hormone therapy for some time, they resumed cross-gender hormone therapy and brain MRI was repeated for comparative reasons. Therefore, we were able to get brain scans of these transgender women at two time points: when they were experiencing hypogonadism (after surgery and not using hormones) and when they were on estrogen replacement therapy. MRI allowed us to obtain measures of both grey matter thickness and connectivity between different brain areas using two techniques. The first of which is called anatomical, because it provides a better macroscopic view of the brain, and the second provides a more functional understanding of the brain. Basically, our aim was to investigate how the cerebral cortex (grey matter) could get thicker or thinner after hypogonadism was corrected and how the connectivity between different brain areas could be influenced by hypogonadism. Additionally, we collected measures of verbal memory and intelligence tests at the two time point.
3) What do findings reveal? How did the brain areas of study patients behave?
Findings may be divided into two types: anatomic and functional. The first showed that some brain areas have a dual behavior during estrogen replacement therapy. This means that, when we corrected hypogonadism and reached low to medium doses of estrogen, the brain “get thick” in some areas; and, when we reached very high doses, these same areas “get thin”. Interestingly, cut-off values to determine low and high levels of estrogen are very similar to estradiol values in the preovulatory follicular phase of the physiological menstrual cycle. Even more interestingly, estradiol has been shown to have the same dual effect on cerebral cortex during the physiological menstrual cycle. As for the second type of finding, as expected from our hypothesis based on brain physiology, there was an increase in connectivity between brain areas related to sensory perception and the so-called “thalamic relay”, which works similarly to an electrical relay. That is, it integrates all brain “electrical inputs” and gathers them for distribution in specific areas of the cerebral cortex. This increase in connectivity seems to show that the “peripheral” perception of sensory stimuli, i.e., heat, tact, pain, and vibration, coming from the body are “better integrated” within the brain when transgender women are using estrogen and their hypogonadism is corrected. Such phenomenon is related not only to sensory stimuli but also to how the sensory perception communicates with the motor “response”, thus being sort of a learning handle that involves sensory and motor portions of the “superior cortical” brain. The above finding also helps to elucidate what happens in menopause: menopause/climateric symptoms occurring after the onset of physiological hypogonadism (ovarian failure) may result exactly from this disruption in the communication between “peripheral” body and brain perception. Additionally, the results for functional analysis, which took a more exploratory approach, showed that the communication between brain areas related to emotional control and depression symptoms is influenced by the correction of hypogonadism.
4) What does information from this research, which was one of the highlights in the recent meeting of the Endocrine Society, represent in view of scientific challenges in the field?
Fortunately, gender-affirming treatment has become more accessible worldwide. The greatest challenge is to promote evidence that benefits transgender women are receiving treatment at an increasingly earlier age, because, unlike menopause, which occurs at a certain age, gender-affirming surgery may be carried out in extremely young people, and we need to better understand which are the “best” ways to manage hypogonadism in these women. And, of course, to fight better against hypogonadism, we need to better understand it. In my opinion, as a specialist in neurosciences, cross-sex hormone therapy should not focus on “mitigating secondary sex characteristics” after gender-affirming surgery, since gonads have already been removed and other procedures, such as mammaplasty, may solve what was initially “solved with estrogen”. I think that the role of estrogen/estradiol in this situation is to serve as hormone replacement, similarly to their role in menopause management. Now there is a growing body of evidence to understand what estradiol does in terms of “neuroplasticity”. This word is used to define the adaptive capacity of brain when facing new situations. Therefore, our study helps to challenge the boundaries of neuroplasticity. New studies may be designed based on ours, and some conclusions may be even extrapolated, with caution, to the non-transgender population, since estrogen is produced locally by the brain, in addition to circulate both in male and female bodies.
5) What is gender dysphoria, how do you characterize patients’ desire, and what can medicine currently offer as an answer?
Gender dysphoria may be summarized as a “strangeness between body and brain”, meaning that people with this condition do not identify themselves with the biological sex to which they were assigned at birth. The biological sex of a person is different from their gender identity. Several studies investigated why brain “perceives” the person’s body as something apart from itself. A growing body of evidence shows that some portions of the so-called “social” and “integrative” brain are different in transgender people compared with non-transgender people. Although it is not possible to state that the brain of a transgender person is identical to that of a person who identifies with the same gender (i.e., the brain of transgender women would be identical to that of non-transgender women), evidence shows that transgender individuals’ brain may present some peculiarities or some of its portions may be somewhere between the brains of men and non-transgender women, in anatomical and functional terms. There are also studies showing that biological and hormone factors, such as “DNA fragments” involved in the translation of hormonal signals, may be different in transgender people. Several theories have been proposed, and those that seem more plausible to me are the ones that argued for a more biological understanding on the expression of gender dysphoria. Patient eagerness for treatment is understandable. The discomfort of being in a body that does not feel like your own is considerable and causes psychological suffering leading to many mental health risks, such as a greater incidence of mood and anxiety disorders. It is known that the number of suicides is much higher (almost 4 times higher) among transgender individuals who do not receive support for their transition/affirmation. All stages of gender affirmation, from social support to hormone therapy and gender-affirming surgery, are associated with improved quality of life and mental health promotion for these people. But this does not mean that all transgender individuals will undergo surgery. Some of them seek for hormone therapy only. It is worth remembering that the definition of gender dysphoria is broad.
6) Which are the next steps of this study? Will this same line of investigation proceed?
These were the first steps. Larger studies are necessary before professionals specialized in transgender health can make full use of our results. Like everything in science, caution should be taken until the establishment of the clinical applicability of these results. For many years, the main research question in the mental health field was the etiology of gender dysphoria (as it is defined in the DSM-5), or gender incongruence (as it is classified in the ICD- 11). Many studies were conducted to investigate “brain” differences that justified the manifestation of gender dysphoria. I believe that there is nothing to explore in terms of etiology. Transexuality is a natural phenomenon, and the only reason for us physicians to intervene is the need of attenuating gender dysphoria, meaning that our goal should be to reduce patients’ suffering by attenuating mismatch with their “peripheral body”. It is well established that transgender individuals are happier when we fit their bodies to their psychological gender than when we tried to “reverse” gender dysphoria. Therefore, our work here as scientists is to ensure that we are providing the best intervention for our patient. And, in this sense, investing the impact of hypogonadism and hormone replacement on mental health allows for the development of better treatment standards and guidelines.
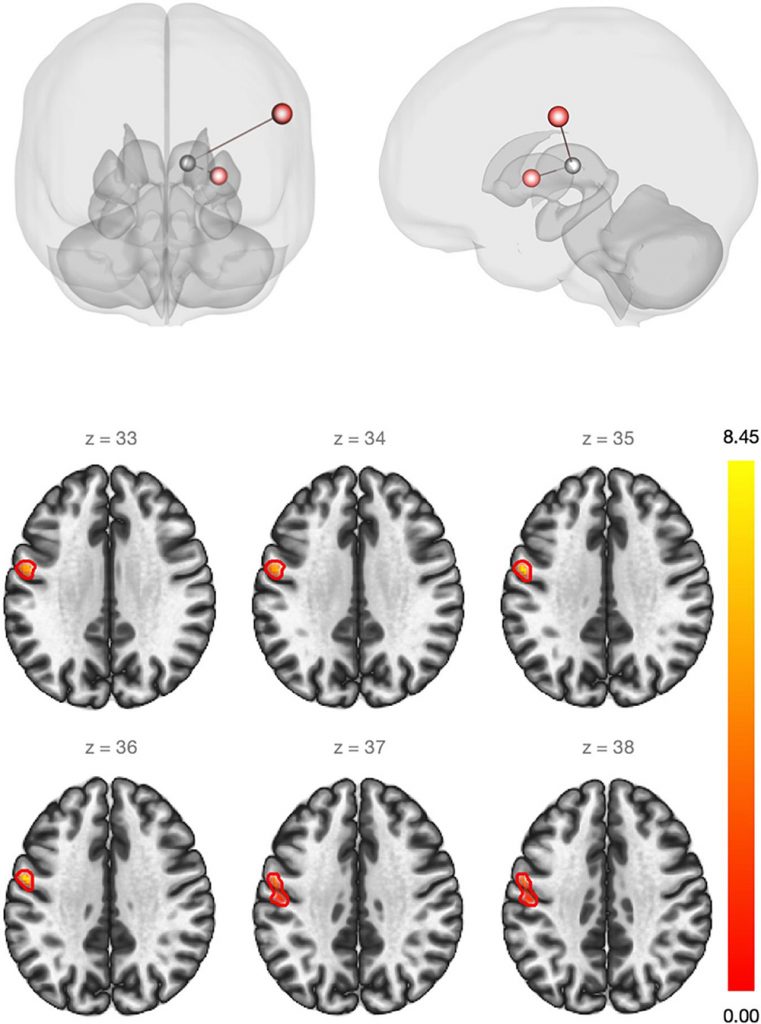
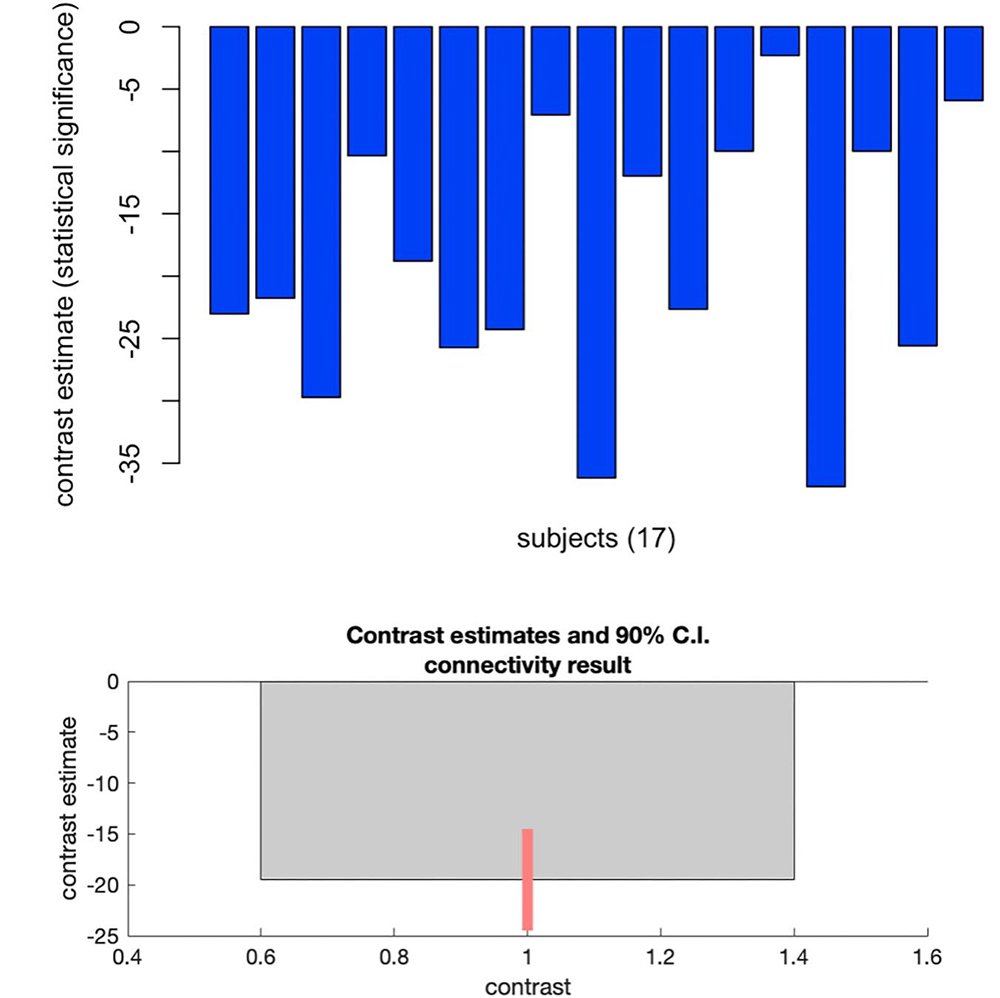
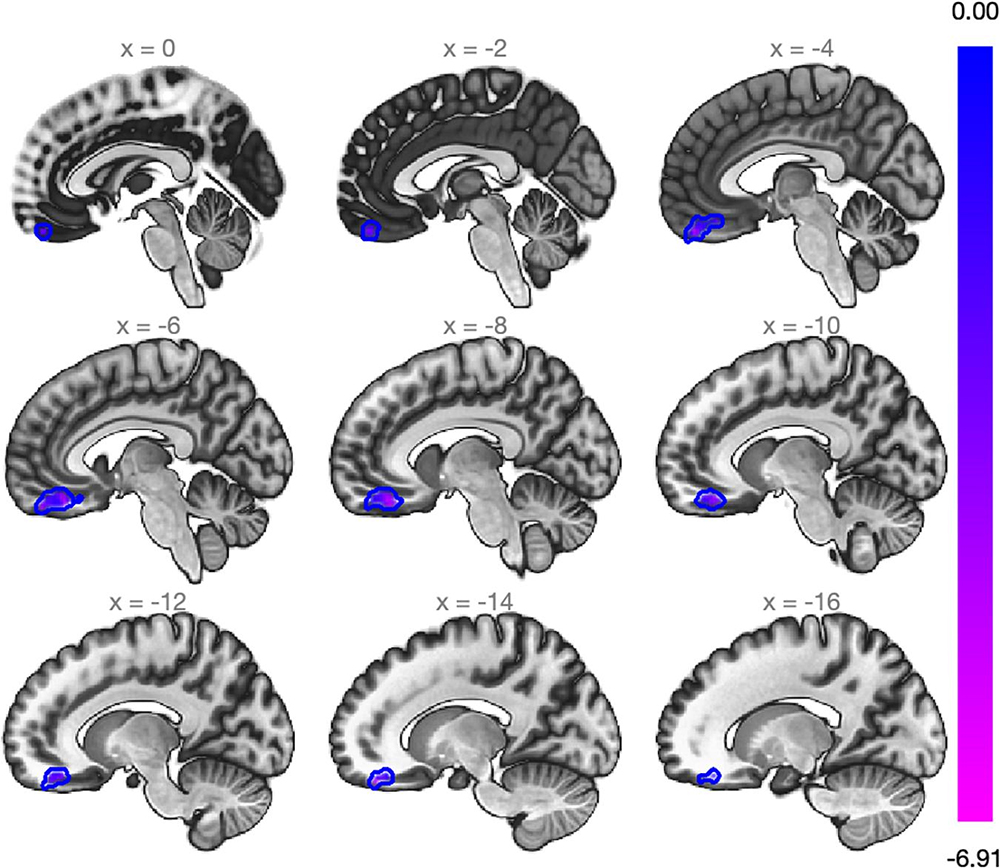
Images reproduced from Frontiers of Neuroscience




